Introduction: Mantle cell lymphoma commonly presents as classic variant histology, however 10-30% patients (pts) can transform into an aggressive histology (blastoid and/or pleomorphic variants). We have previously reported that genomic profile and clinical outcomes of transformed MCL pts are distinct from classic variant pts who never transformed (CNT) and that transformed pts have poor clinical outcomes. In this study, we analyzed the factors at initial diagnosis which can predict the time to transformation and the risk for transformation in a cohort of 369 pts with MCL.
Methods: We analyzed charts from 369 pts with MCL (293 were CNT and 76 were transformed MCL). Statistical analysis was performed from baseline pt characteristics collected from the time of initial diagnosis in CNT group and at the time of initial diagnosis of classic variant MCL who later transformed (t-MCL). Time to transformation (TTT) was calculated from initial diagnosis to the date of transformation in those who transformed and last follow up in those who never transformed. Univariate and multivariate (MVA) logistic regression modeled the risk of transformation. Classification and regression tree (CART) analysis was performed to identify optimal cut off in categorical variables predictive of TTT.
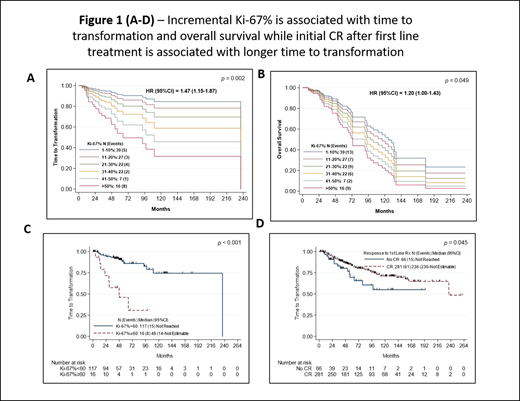
Results: Among the 369 pts, the median age was 62 yrs (range 34-90). Ki-67% values were available in 133 pts (36% of total) and median Ki-67% was 25% (range 1-80). Three hundred eight pts (84%) had initial bone marrow involvement and 66 pts (18%) had leukemic phase at diagnosis. The median follow up was 133 months and the median overall survival (OS) was 95 months and 43% were alive at the time of this analysis. Discernible difference were noted in pts who belong to t-MCL group compared to pts in the CNT group, pts in t-MCL group exhibited - higher values of median Ki-67% (30% vs 20% in CNT; p=0.04), higher LDH levels, higher proportions of pts with high risk simplified MIPI risk score, leukemic phase at initial diagnosis, complex karyotype and lower hemoglobin. First line treatments received by both groups were similar. Eleven pts got anti-CD19 CART therapy at some point and 7 pts were t-MCL and 4 were CNT group. The median time to transformation in months for those who transformed was 39 (range 5-240 months) while in CNT it was 51 months (1-257 months). We further identified that incremental Ki-67% was significantly associated with TTT and OS (Figure-1A-B). Using CART analysis we identified Ki-67% ≥60 is significantly associated with shorter TTT (HR, 6.26; 95% CI 2.58-15.21; p <0.001-1C). Shorter TTT was associated with elevated LDH, high risk MIPI score, CNS involvement at baseline, higher WBC counts, leukemic phase MCL, complex cytogenetics and R-chemotherapy as first line treatment. Longer TTT was associated with higher hemoglobin, CR after initial treatment (Figure-1D) and increased number of lines of treatment before transformation. In MVA, CNS involvement at baseline, bone marrow involvement, first line R-chemotherapy, high LDH (>729 IU/L), higher platelet count > 180 and high absolute lymphocyte count > 5000 k/ul were predictive of shorter TTT.
Logistic regression model showed factors associated with the risk of transformation. In univariate analysis, higher risk was significantly associated with Ki-67% as a continuous variable - OR 1.03 (95% CI 1.01-1.05; p=0.006), leukemic phase at diagnosis, high risk MIPI score and complex cytogenetics. First line treatment with ibrutinib compared to R-HCVAD, was associated with decreased risk of transformation. In MVA, ibrutinib/BTK inhibitor therapy (n=51) as first line therapy was associated with decreased risk of transformation while CNS involvement was associated with higher risk of transformation.
Conclusions: Routinely available clinical variables can help determine the risk for transformation of MCL and time to transformation. Progressive increase in Ki-67 and high MIPI risk score is associated with shorter TTT and increases the risk for transformation. Earlier usage of BTK inhibitor therapy may reduce the risk of developing histologic transformation in MCL pts.
Lee:Seattle Genetics: Research Funding; Takeda: Research Funding; Aptitude Health: Speakers Bureau; Oncternal Therapeutics: Research Funding; Guidepoint Blogal: Consultancy; Celgene: Research Funding; Bristol-Myers Squibb: Consultancy, Research Funding. Vega:NCI: Research Funding. Flowers:Celgene: Consultancy, Research Funding; Karyopharm: Consultancy; Millennium/Takeda: Consultancy, Research Funding; Acerta: Research Funding; Spectrum: Consultancy; Pharmacyclics/Janssen: Consultancy; Leukemia and Lymphoma Society: Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Research Funding; OptumRx: Consultancy; TG Therapeutics: Research Funding; BeiGene: Consultancy; Denovo Biopharma: Consultancy; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; Cancer Prevention and Research Institute of Texas: Research Funding; Burroughs Wellcome Fund: Research Funding; Eastern Cooperative Oncology Group: Research Funding; National Cancer Institute: Research Funding; V Foundation: Research Funding; Kite: Research Funding; Bayer: Consultancy. Wang:Kite Pharma: Consultancy, Other: Travel, accommodation, expenses, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding; Janssen: Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding; Beijing Medical Award Foundation: Honoraria; Dava Oncology: Honoraria; Molecular Templates: Research Funding; Verastem: Research Funding; InnoCare: Consultancy; Nobel Insights: Consultancy; OncLive: Honoraria; Loxo Oncology: Consultancy, Research Funding; MoreHealth: Consultancy; Oncternal: Consultancy, Research Funding; Targeted Oncology: Honoraria; Acerta Pharma: Research Funding; Guidepoint Global: Consultancy; Pharmacyclics: Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding; Pulse Biosciences: Consultancy; Lu Daopei Medical Group: Honoraria; Juno: Consultancy, Research Funding; VelosBio: Research Funding; BioInvent: Research Funding; OMI: Honoraria, Other: Travel, accommodation, expenses; Celgene: Consultancy, Other: Travel, accommodation, expenses, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal